Recent results indicate that chronic HBV and HCV infections, respectively, have a suppressive effect on host immune responses. Such observations call for complementary host-directed biological therapies, which are able to stimulate the native immune response restoring the host natural immune responsiveness against viruses. HepC – Superinfection Therapy (SIT), developed by HepC – Superinfection, is such a modality.
HepC – Superinfection has selected IBDV, a non-pathogenic double stranded RNA (dsRNA) virus, a strong activator of antiviral gene responses as its primary vector system for product development. The Company believes that IBDV will be the first vector systems to receive regulatory product approval employing the innovative SIT concept, because of its superior delivery characteristics, its safety profile and its efficacy for clinical applications with unmet medical need. This way, HepC – Superinfection is focusing on three key product areas making use of its IBDV platform technology:
- Functional cure of advanced chronic HBV during a finite treatment course; chronic HBV patients currently require life-long treatment with available NUC drug candidates (Lead product HC001). Achieving a functional cure, the development of HCC would also be prevented in these patients.
- In HBV/HCV coinfected individuals, when HCV infection is suppressed by DAA agents and their HBV can reactivate. We wish to treat HBV/HCV coinfected patients without reactivation of HBV (Second product HC002).
- As interferon is active against most vertebrate-infecting viruses, SIT could also mitigate the death toll of the next flu pandemic by an unconventional viral interference strategy, which aims to complement the development of universal prophylactic flu vaccines. Since our drug candidate is easy-to-produce and store, it could generate financial incentive for the development of a post-infection pandemic therapeutic flu vaccine (Third product HC003).
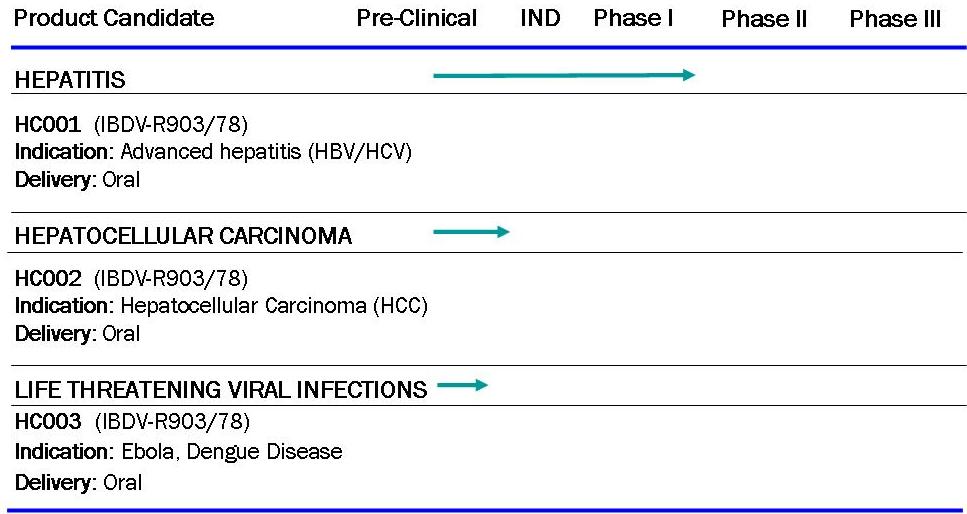
Three key product areas making use of the IBDV platform technology.
